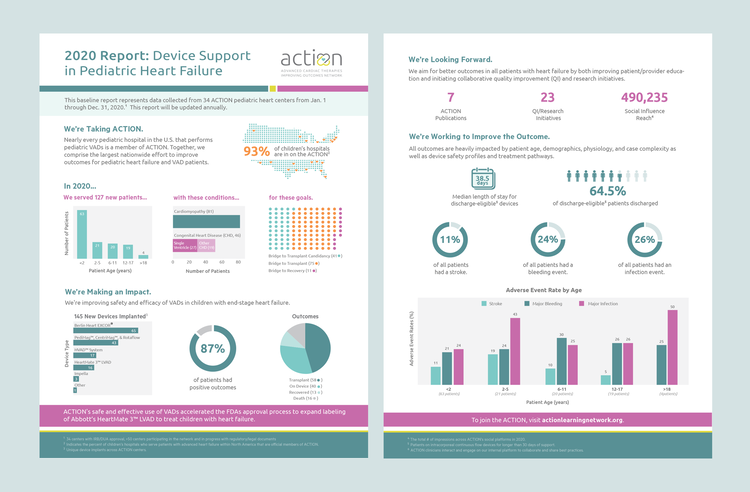
Berlin Heart EXCOR and ACTION Post-Approval Surveillance Study Report
Cerebrovascular accidents (CVA) are a common problem in pediatric VAD support. This Pedimacs analysis investigates rates, risk factors, and outcomes The field of pediatric mechanical circulatory support changed dramatically when the Berlin Heart EXCOR® VAD was FDA-approved for use in children nearly two decades ago. This ACTION publication describes the outcomes of patients with a Berlin […]
Berlin Heart EXCOR and ACTION Post-Approval Surveillance Study Report Read More »