Heart of ACTION

NEW! Heart Failure Success Patient & Family Education Handbook
ACTION is excited to release our first patient and family education handbook focused on Heart Failure Success!
Latest Highlights

NEW! Heart Failure Success Patient & Family Education Handbook
ACTION is excited to release our first patient and family education handbook focused on Heart Failure Success!

The Pulse — July 2024
Dear Community, We hope you are enjoying your summer vacations and finding time to rest and rejuvenate with your families! ACTION has been busy with new publications, tools, and resources. Special thanks to the Muscular Dystrophy & Heart Failure committees for new harmonization protocols, and to the Fontan Referral Improvement Project leadership & committee for launching the Provider Grand Rounds deck for Advanced Therapeutic Options for Fontan Patients!

A new HeartMate 3™ LVAD Patient Education Handbook for ADULTS is Now Available!
ACTION is proud to release our newest patient education material for ADULT patients with the HeartMate 3™ LVAD.

Mechanical Ventilation and Outcomes of Children Who Undergo Ventricular Assist Device Placement: 2014–2020 Linked Analysis From the Advanced Cardiac Therapies Improving Outcomes Network and Pediatric Cardiac Critical Care Consortium Registries
Placement of a ventricular assist device (VAD) improves outcomes in children with advanced heart failure, but adverse events remain important consequences. Preoperative mechanical ventilation (MV) increases mortality, but it is unknown what impact prolonged postoperative MV has.
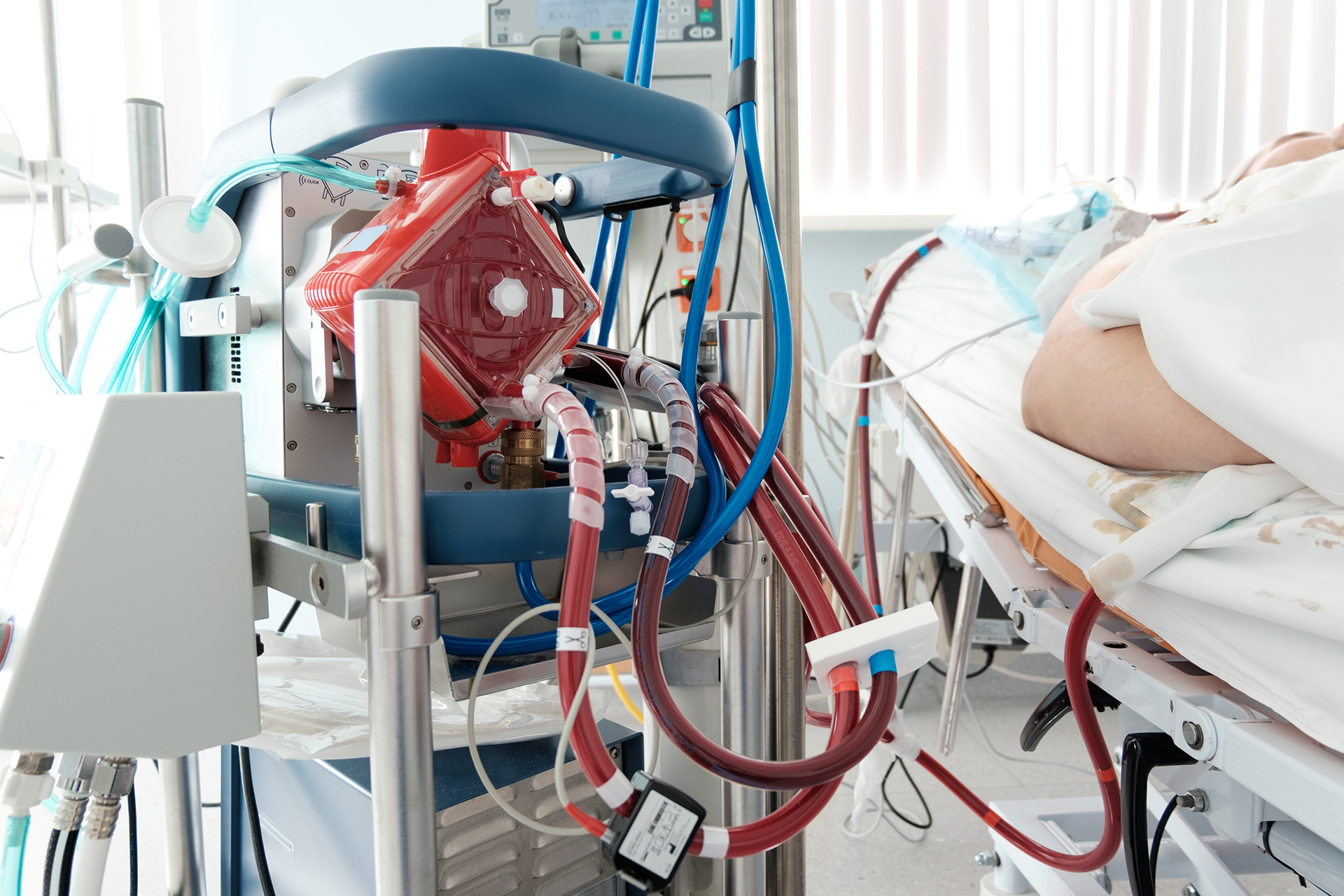
Transcatheter Axial Pump Use in Pediatric Patients on Veno-Arterial Extracorporeal Membrane Oxygenation: An ACTION Collaborative Experience
Abstract We report the largest pediatric multicenter experience with Impella pump use and peripheral veno-arterial extracorporeal membrane oxygenation (VA-ECMO) support. Utilizing the Advanced Cardiac Therapies Improving Outcomes Network (ACTION) collaborative
SEE ALL
